As a manufacturer of medical suction tubes and other medical devices in the contract manufacturing sector, we bear a special responsibility for the safety of patients and users. Therefore, we consistently align all our processes with the stringent requirements of ISO 13485 and MDR 2017/745 (Medical Device Regulation)
From development through production to delivery, our products are subject to strict quality controls. Carefully selected materials, qualified employees and standardised testing procedures ensure consistently high product quality.
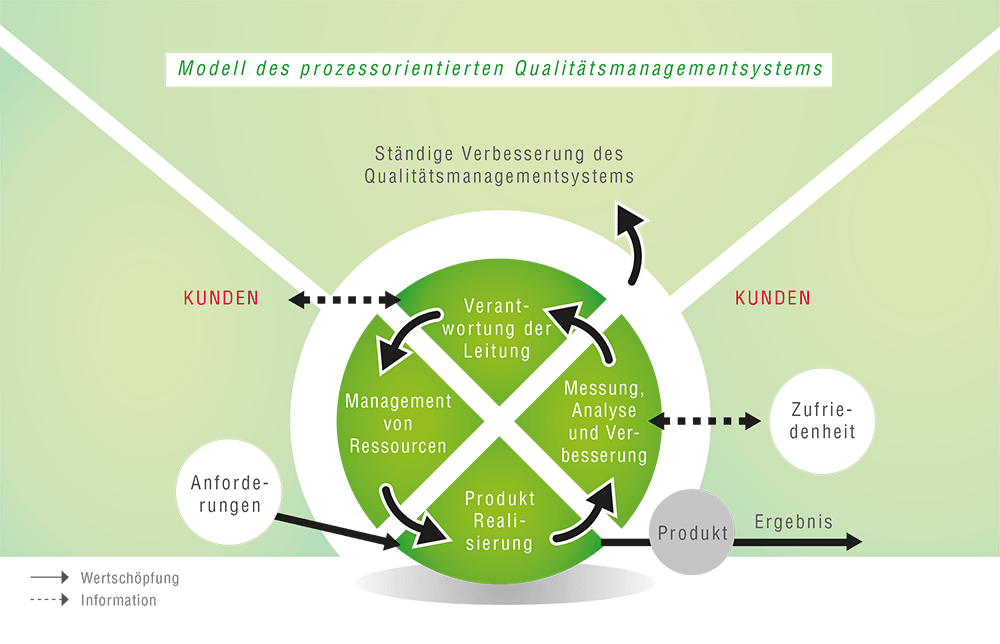
Our certified quality management system is regularly reviewed and continuously improved. This ensures that our suction tubing not only complies with legal requirements but also meet the highest standards of functionality, reliability and safety.
Certification according to ISO 13485 and MDR 2017/745 (Medical Device Regulation) confirms our commitment to the highest quality and safety standards in the manufacture of medical devices.

